Color Trading Sp. z o. o. Asia-Pacific site
- English
Please select your Region.
Please select your Region.
Aug 31, 2017
Detects trace of virus at early stage by highly sensitive rapid measurement.
- Early determination in one and a half minutes (fastest) is possible for influenza diagnosis while simultaneous multiple sample measurement is also available. -
The influenza virus spreads predominantly in the winter season of each year, and many people suffer from this acute viral infection. Serious cases of the illness carry the risk of pneumonia in the elderly, and may cause complications of encephalitis and encephalopathy in small children. In order to avoid those serious cases, complications and secondary infection, rapid testing at the early stage, early diagnosis and prompt commencement of treatment are necessary.
Diagnostic reagents using the immunochromatography method have been widely used in medical facilities. In this method, an antigen-antibody reaction caused by mixing a sample which is taken from the patient's nose or throat into the reagent creates a line on the reagent, and a positive/negative determination will be made based on the line. While this method is easy and convenient, making a positive diagnosis is difficult unless the amount of virus increases to a certain level and a margin of error tends to occur due to visual judgment. Therefore, automatic determination by devices has been getting popular. However, since the current available devices can only measure and give a result for one sample at a time, handling many samples during the flu season is difficult. Hence the development of a highly sensitive device which measures multiple samples simultaneously had been longed for.
ARKRAY, Inc. (hereafter 'ARKRAY') will launch SPOTCHEM FLORA SF-5520 which uses the TRF (Time Resolved Fluorescence) method and enables highly sensitive simultaneous measurement for multiple samples. For influenza testing, the dedicated reagent SPOTCHEM FLORA FluAB using europium, which has a fluorescence life that is 0.2 million times longer than normal fluorescent substances, is available. With this reagent, more sensitive fluorometric measurement that is free from the effect of excitation light (noise) is possible, in turn enabling detection and measurement at the early stage where the amount of virus is small. In addition to simultaneous measurement of three samples at a maximum, the instrument is equipped with a function allowing users to measure additional samples at any time even when another measurement is ongoing. Thanks to this function, the influenza virus can be detected easily, conveniently and efficiently during the flu season.
ARKRAY shall continue contributing to rapid diagnosis and appropriate treatment in the front line of the medical field through the development of new diagnosis systems.
• High detection performance
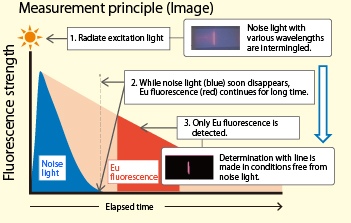
・High detection performance is realized through the TRF method employing europium (Eu) as a labeling substance, which has a long fluorescence life and fluorescence wavelength that allows for easy detection of the virus.
・In the field of influenza testing, making a positive judgment is currently difficult unless the amount of virus in the body increases to a certain level.
・ However, highly sensitive detection even at the early stage with a small amount of virus has been achieved, contributing to the reduced risk of epidemics because of the commencement of early treatment.
• Reduction in testing time by early determination*
Highly sensitive determination is made four times in total, starting from one and a half minutes (fastest, followed by 2.5 min., 4 min. and 6 min.) and if a positive reaction is detected, an early determination result is printed out before the final determination is made (10 min.). This contributes to reducing the burden on patients by shortening testing time and waiting time.
*Function at time of influenza virus antigen measurement
• Multiple sample measurement
In addition to simultaneous measurement of three samples at a maximum, even when a measurement is ongoing, measurements for additional samples are possible at any time. Measurements during the flu season can be performed efficiently.
• Simple and automatic measurement
By simply inserting a cartridge to which pre-processed sample is dripped into the instrument, the result is automatically determined. Visual determination or time management conducted by a person is no longer necessary.
• Improvement in operation
Equipped with a color touchscreen display and LED lamp, SF-5520 allows users to easily monitor testing progress at any time.
• Prevention against mix-up of samples
Names and symbols can be written on cartridges. The instrument reads them along with the test results, which helps to prevent the mix-up of samples.
• On-line operation is possible
On-line operation with a hand-held barcode reader (separately sold) is possible, resulting in safe and efficient operation even during the flu season or in facilities where measurement volume is high.

The TRF method employing europium (Eu) as a labeling substance, which has a long fluorescence life and fluorescence wavelength that allows for easy detection of the virus, is adopted. Fluorometric measurement without the influence of excitation light (noise) enables the instrument to detect much weaker reactions. By employing this principle, highly sensitive detection has been achieved even for a small volume of antigen at the early stage.


| Name | Portable Immunofluorometric Analyzer SPOTCHEM FLORA SF-5520 | |
|---|---|---|
| Specifications | ||
| Test item | Influenza virus antigen (type A/B), Group A Streptococcus antigen | |
| Measurement principle | TRF method (Time Resolved Fluorescence method) | |
| Reaction timet | Influenza antigen: 10 min. (early determination function equipped) Group A Streptococcus antigen: 5 min. |
|
| No. of loadable samples | Three samples | |
| Measurement modes | Automatic measurement mode/Scan mode | |
| Built-in printer | 58mm thermal printer | |
| Data memory | 1000 samples | |
| Measurement environment | Temperature: 15℃ to 30℃ Humidity: RH20% to 80% (no condensation) | |
| Dimensions | 290mm (W) × 210mm (D) × 200mm (H) | |
| Weight | 2.2kg | |
| Power source | AC adapter (100V to 240V, 50/60Hz) | |
This product will be sold through ARKRAY Marketing, Inc.
ARKRAY Marketing, Inc. is ARKRAY's distributor in Japan.
Sales except Japan to be determined.
![]()